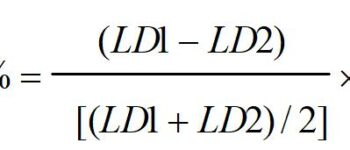Introduction
Reasonable and effective utilization of coal or biomass is an important goal of coal/biomass chemical engineering at present and in the future. Among the possible technical routes, ethylene glycol (EG), which is widely used as an important chemical in polyester manufactures or as antifreeze and so forth,1 can be synthesized from syngas derived from coal and biomass gasification instead of the traditional manufacture from petroleum.2
As is well known to all, supported metal catalysts with multicomponents are among the most commonly used catalysts because of their obvious advantages such as the enhanced activity and stability. Compared to the traditional support (silica, alumina, or titania),3−5zirconia is frequently employed as a preferable support for Cu-based catalysts in hydrogenation reactions due to its ability to easily form strong metal-support interaction, which can produce distinctive active phases especially the Cu-ZrO2 interface.6−8 As a consequence, CuO-ZrO2 catalysts are considered to exhibit excellent catalytic performance in EG synthesis.6,9 Unfortunately, many drawbacks such as low selectivity and poor thermal stability in hydrogenation reactions are brought by the intrinsic disadvantages of traditional Cu-based catalysts. The deficiency in CuO-ZrO2 catalysts calls for a better solution to meet the tough challenges, which is also at the very heart of fabricating high-performance catalysts for EG synthesis. Regarding the extensively studied CuO-ZrO2 catalysts, the interaction between Cu species and support is generally believed to be pivotal in enhancing selectivity and stability.10 Research is therefore focused on the development of advance synthetic methods with high uniformity in multicomponent distribution or focused on the addition of promoters.11 For the former, the simple one-pot complexation route offers flexibility in design to fabricate multicomponent catalysts. As a result, the obtained catalysts have higher uniformity and tightness in combination of various components, resulting in the enhanced synergistic effects in catalysis reaction.8,12 With regard to the latter strategy, various promoters are added to obtain the functional CuO-ZrO2 catalysts with desired physical and chemical properties.13−15 The introduction of La into Cu-based catalyst frequently changes the electron density of metal active sites, which thus enhances their catalytic activity and stability in hydrogenation reactions.16 To be honest, there is still some controversy about the influence of preparation conditions such as the calcination temperature on the complexation route. Hence, to find the appropriate calcination temperature is a precondition for catalyst preparation, which can modify the structural properties, avoid heterogeneous distribution of metal species, and strengthen their interaction with each other.
Bạn đang xem: Enhanced Ethylene Glycol Selectivity of CuO–La2O3/ZrO2 Catalyst: The Role of Calcination Temperatures
In this paper, the La-doping CuO-ZrO2 catalysts prepared by a simple one-pot salicylic complex process have been applied to selectively synthesize EG by hydrogenation of diethyl oxalate (DEO) or dimethyl oxalate (DMO). Effects of calcination temperatures on the structure and activity of CuO-La2O3/ZrO2 catalyst were investigated, and the optimal temperature was found out. All the catalysts were characterized by N2-physisorption, chemisorption, and other techniques to study their structural features and catalytic properties. Combined with the activity evaluation results in hydrogenation of oxalates to EG, the CuO-La2O3/ZrO2 catalyst calcined at a higher temperature (700 °C) could afford a much higher selectivity and good yield to EG.
Nguồn: https://thuvienhaichau.edu.vn
Danh mục: Hóa