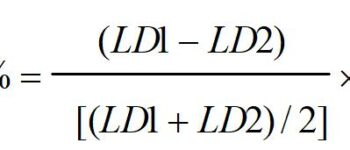1. Introduction
Layers of metal atoms occur in the mesosphere-lower thermosphere (MLT) region of the Earth’s atmosphere as a result of the ablation of cosmic dust particles. The total input of this dust has recently been estimated to be 43 ± 14 tonnes day-1, of which around 92% comes from comets and the rest comes from the asteroid belt between Mars and Jupiter.1 The ablated metal atoms occur in layers between 80 and 105 km, which are global in extent. Four neutral metal atoms, Na, Fe, K, and Ca, can be observed from the Earth’s surface using the resonant lidar technique; uniquely for ground-based observations, the Ca+ ion layer can also be observed and occurs ∼5 km above the neutral Ca layer.2 Although Ca has a similar elemental abundance to Na in meteorites, the Ca atom abundance in the MLT is roughly 2 orders of magnitude smaller than that of Na.3−5 One reason for this Ca depletion is the highly refractory nature of CaO in molten meteoroids,6 so that Ca ablates about 1 order of magnitude less efficiently than Na.1 We have recently confirmed this differential ablation using a novel meteoric ablation simulator.7
A second reason for the Ca depletion may be that Ca is converted to very stable reservoir species, which are less easy to recycle back to atomic Ca than the corresponding Na reservoir, NaHCO3.8 A good understanding of the cycling between elemental Ca and its reservoirs, including Ca-bearing ions, is emerging.9−11 However, there is still a great deal of uncertainty about the nature of the stable Ca reservoirs. This is important because the formation of these reservoirs governs the lower ledge of meteoric atomic layers and, in some cases, may even determine the seasonal behavior of the layer as a whole.12 Also, these processes determine the way in which metals incorporate into meteoric smoke particles (MSPs), which are nanometer-sized particles that form from the condensation of metallic species in the MLT.2
Bạn đang xem: Reaction Kinetics of CaOH with H and O2 and O2CaOH with O: Implications for the Atmospheric Chemistry of Meteoric Calcium
Figure Figure11 is a schematic diagram of calcium chemistry in the MLT. Ca atoms react with O3 to produce CaO13
which can then be recycled back to Ca by reaction with O(3P) or further react to yield reservoir molecules.10,14
Although Ca(OH)2 is thermodynamically very stable, it reacts rapidly with H atoms to yield CaOH.10The role of the CaOH radical as a reservoir species is the focus of the present study. CaOH can recycle back to Ca via a reaction that is well-known in the field of metal-catalyzed flame chemistry
15,16
where the combination of reactions R2a, R5, and R6b in essence catalyzes the recombination of 2H atoms to form H2.
The reaction enthalpies at 0 K for reactions R1, R2a, R5, and R6 have been calculated using available heats of formation taken from data evaluations.17−19 The preferred heats of formation of CaO and CaOH at 0 K (Figure S1 of the Supporting Information) are 26 ± 17 kJ mol-1 19 and −170 ± 15 kJ mol-1,17 respectively. Enthalpies of reaction for reactions R3 and R4 were obtained from electronic structure calculations at the B3LYP/6-311+(2d,p) level of theory (estimated uncertainty of ±20 kJ mol-1).10,14
Xem thêm : Offenes System: Austausch von CO2 und O2
As shown in Figure Figure11, an unexplored and potentially important fate for CaOH could be its association with O2 [enthalpies of reaction from B3LYP/6-311+(2d,p) calculations can be found in the Supporting Information].
The O2CaOH molecule can then recycle CaOH back by reaction with O(3P) in two steps
or associate with other metal-containing molecules to generate MSPs.
The reaction of CaOH and O may also be slightly exothermic and could potentially recycle reactive calcium.
In this paper, we investigate the atmospheric fate of CaOH by studying experimentally the reactions of CaOH with H and O2 and the recycling of CaOH from O atom reactions.
Nguồn: https://thuvienhaichau.edu.vn
Danh mục: Hóa